FDA Approves Wegovy Weight-Loss PillFDA Approves Wegovy Weight-Loss Pill

(MedPage Today) — The FDA approved a pill version of the GLP-1 receptor agonist semaglutide (Wegovy), making it the first oral GLP-1 agent indicated for weight loss, Novo Nordisk announced on Monday. The once-daily 25 mg pill is approved to lower… (MedPage Today) — The FDA approved a pill version of the GLP-1 receptor Read More
FDA Approves Pill Version of Wegovy: What You Need to KnowFDA Approves Pill Version of Wegovy: What You Need to Know
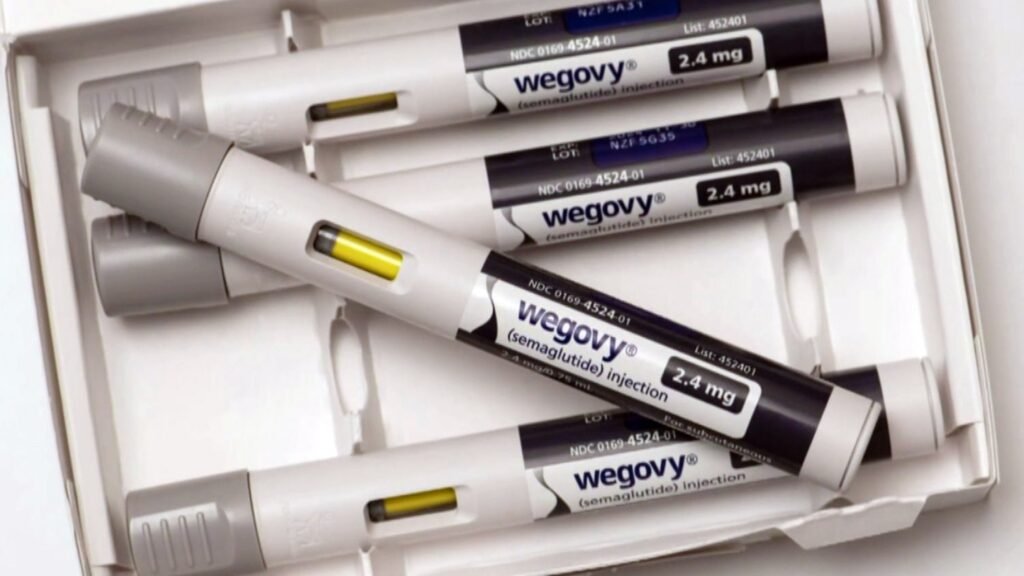
The Food and Drug Administration has approved the popular weight loss drug Wegovy in pill form after it had only been available as an injection. NBC News medical contributor Dr. Natalie Azar joins TODAY to break down the effectiveness of it, potential side effects, cost and more. The Food and Drug Administration has approved the Read More
U.S. regulators approve Wegovy pill for weight lossU.S. regulators approve Wegovy pill for weight loss

U.S. regulators on Monday gave the green light to a pill version of the blockbuster weight-loss drug Wegovy, the first daily oral medication to treat obesity. (Image credit: Jacquelyn Martin) U.S. regulators on Monday gave the green light to a pill version of the blockbuster weight-loss drug Wegovy, the first daily oral medication to Read More
Ozempic Meals? Restaurants shrink portions to match bite-sized hungerOzempic Meals? Restaurants shrink portions to match bite-sized hunger

A self-described foodie, social butterfly and New Yorker for 20 years, Lina Axmacher has long loved exploring the city’s famed restaurant culture. A self-described foodie, social butterfly and New Yorker for 20 years, Lina Axmacher has long loved exploring the city’s famed restaurant culture.
Novo Nordisk says US approves first GLP-1 pill for weight lossNovo Nordisk says US approves first GLP-1 pill for weight loss

Danish pharmaceutical giant Novo Nordisk announced Monday the US Food and Drug Administration (FDA) had approved its popular GLP-1 anti-obesity drug Wegovy to be administered in pill form for weight loss. Danish pharmaceutical giant Novo Nordisk announced Monday the US Food and Drug Administration (FDA) had approved its popular GLP-1 anti-obesity drug Wegovy to be Read More
US regulator approves pill form of Wegovy weight-loss drugUS regulator approves pill form of Wegovy weight-loss drug
Wegovy becomes first pill of its kind to be approved, shifting weight-loss drugs beyond injections. Wegovy becomes first pill of its kind to be approved, shifting weight-loss drugs beyond injections.
US regulators approve Wegovy pill, first oral medication to treat obesityUS regulators approve Wegovy pill, first oral medication to treat obesity
Food and Drug Administration’s approval hands drugmaker Novo Nordisk an edge in the race to market an obesity pill US regulators on Monday gave the green light to a pill version of the blockbuster weight-loss drug Wegovy, the first daily oral medication to treat obesity. The US Food and Drug Administration’s approval handed drugmaker Novo Read More
FDA approves Wegovy pill for weight lossFDA approves Wegovy pill for weight loss

The Food and Drug Administration has approved a pill version of the weight-loss drug Wegovy. The Food and Drug Administration has approved a pill version of the weight-loss drug Wegovy.
Gestational Weight Gain and Pregnancy Outcomes After GLP-1 Receptor Agonist DiscontinuationGestational Weight Gain and Pregnancy Outcomes After GLP-1 Receptor Agonist Discontinuation
This cohort study examines gestational weight gain and pregnancy outcomes after glucagon-like peptide-1 (GLP-1) receptor agonist discontinuation. This cohort study examines gestational weight gain and pregnancy outcomes after glucagon-like peptide-1 (GLP-1) receptor agonist discontinuation.
Increasing Postpartum Use of GLP-1 Receptor AgonistsIncreasing Postpartum Use of GLP-1 Receptor Agonists
This study uses data from the Danish Medical Birth Register to examine postpartum use of glucagon-like peptide 1 receptor agonists. This study uses data from the Danish Medical Birth Register to examine postpartum use of glucagon-like peptide 1 receptor agonists.